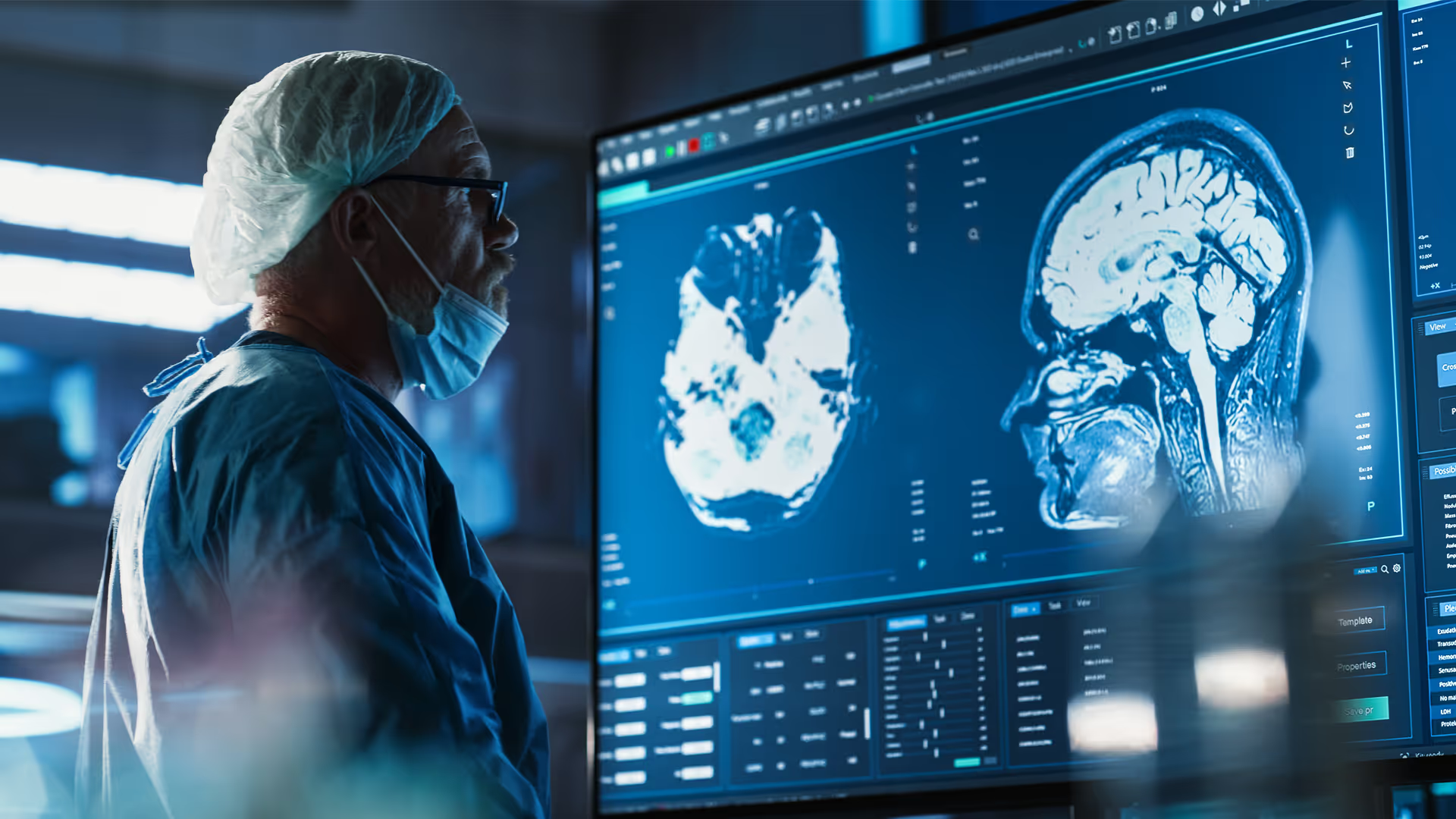
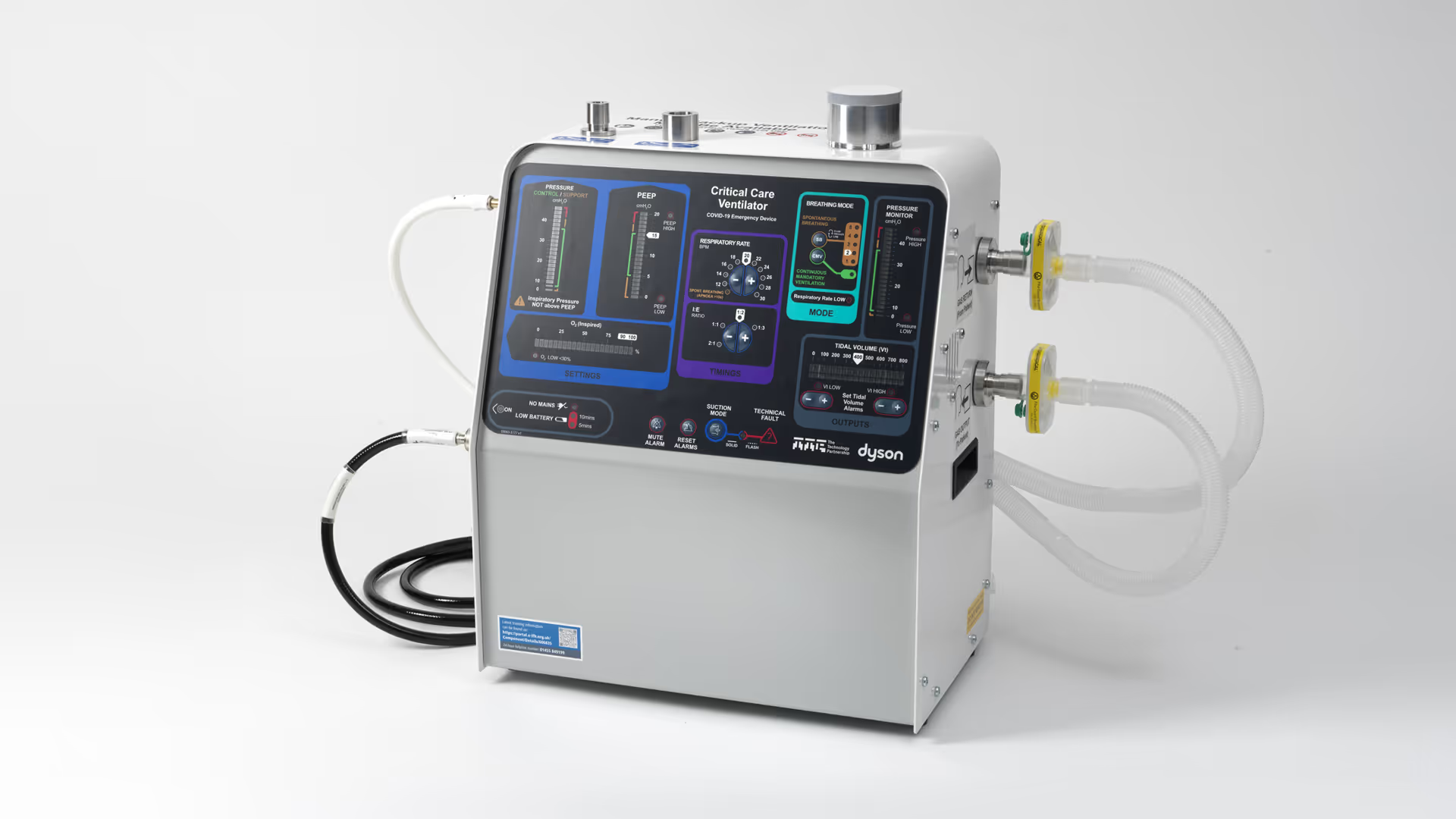
Cardiovascular device development
Solving the toughest challenges in cardiovascular innovation.
With decades of experience advancing medical technologies, we help innovators design, develop, and commercialise the next generation of cardiovascular devices. Our multidisciplinary teams combine engineering, science, and human factors expertise with capabilities in robotic-assisted interventions, advanced imaging, algorithms, and digital modelling to tackle complexity and accelerate innovation. From structural heart to rhythm management, we deliver technologies that improve patient outcomes, meet clinical needs, and achieve commercial success.


Structural heart interventions
Designing technologies for structural heart interventions requires navigating complex anatomical, physiological, and regulatory challenges.
At TTP, we apply first-principles thinking, advanced modelling and systems engineering for next-generation solutions - delivering safe, reliable, and high-performing devices across prosthetic valves, repair systems and occlusion technologies for our clients.
We work closely with clients at the forefront of transcatheter intervention, supporting the development of robotic-assisted systems, fusion imaging platforms, and smart implants with embedded sensing and connectivity.
These technologies are enabling greater standardisation of complex procedures, reducing clinician cognitive burden, and improving procedural precision and the consistency of clinical outcomes – while also supporting shorter learning curves and new models of training and supervision, such as teleproctoring.

TTP has been creative, thorough and quick. We now have a much better solution to meet the needs of our unique operational environment.
Dwight Meglan
Chief Technology Officer
,
Heartlander Surgical

I’ve had the opportunity to work closely with TTP over the past 20 years, focused on solving very diverse and complex engineering challenges, and required to deliver breakthrough product technology to the market. They’ve been incredible partners and fully integrated members of our R&D team and have shared tremendous passion, energy, and drive to succeed to help support our physicians and their patients.
Joshua Stopek
VP: R&D
,
HistoSonics Inc

We gave TTP an underdefined, complex therapy characterisation challenge in a novel therapy space. They, in turn, provided a systematic discovery plan, delivered rigorous and understandable modelling and neuroscience outputs, and ultimately steered us towards a workable concept for a new-to-the-world therapy concept. I was reliably impressed with their responsiveness, the quality of the outputs, and their ability to iterate at high velocity towards answers to really hard questions.
Steven Goetz
VP Technology & Platform Innovation
,
Medtronic Neurom.
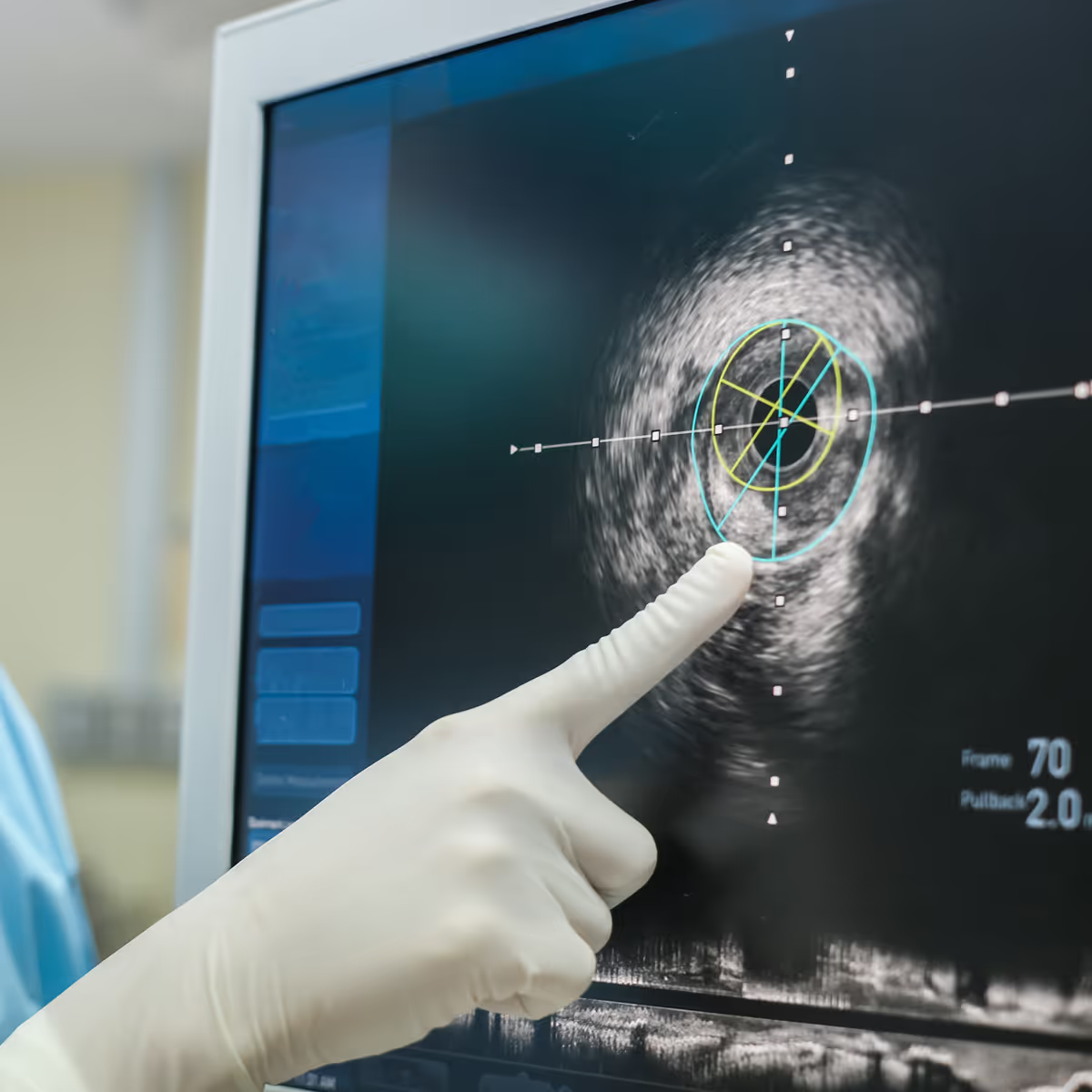

Peripheral and neurovascular interventions
From peripheral artery disease to acute ischemic stroke, TTP supports the development of endovascular devices that restore blood flow and improve patient recovery. Our expertise spans fluidic management systems, stent and implant delivery platforms, thrombectomy and embolic protection devices, and innovative catheter-based solutions.
We integrate fusion imaging and navigation technologies to improve precision across cath lab and neuro interventional or hybrid operating environments. Looking ahead, we are enabling hybrid approaches that integrate mechanical intervention with non-implant based energy therapies, reducing procedure time and improving long-term outcomes.


Cardiac rhythm management and pacing
Pacemakers, defibrillators, and emerging leadless systems demand miniaturisation, power efficiency, and uncompromising reliability. We help clients advance these systems with low-power electronics, wireless telemetry, smart algorithms and biocompatible materials.
In the electrophysiology lab, we support the development of technologies that integrate algorithms, simulations, and digital twin models to optimise device performance and guide therapy.
We also explore remote monitoring solutions that extend the value of pacing and rhythm devices beyond the procedure, enabling clinicians to track patient health and device function continuously.
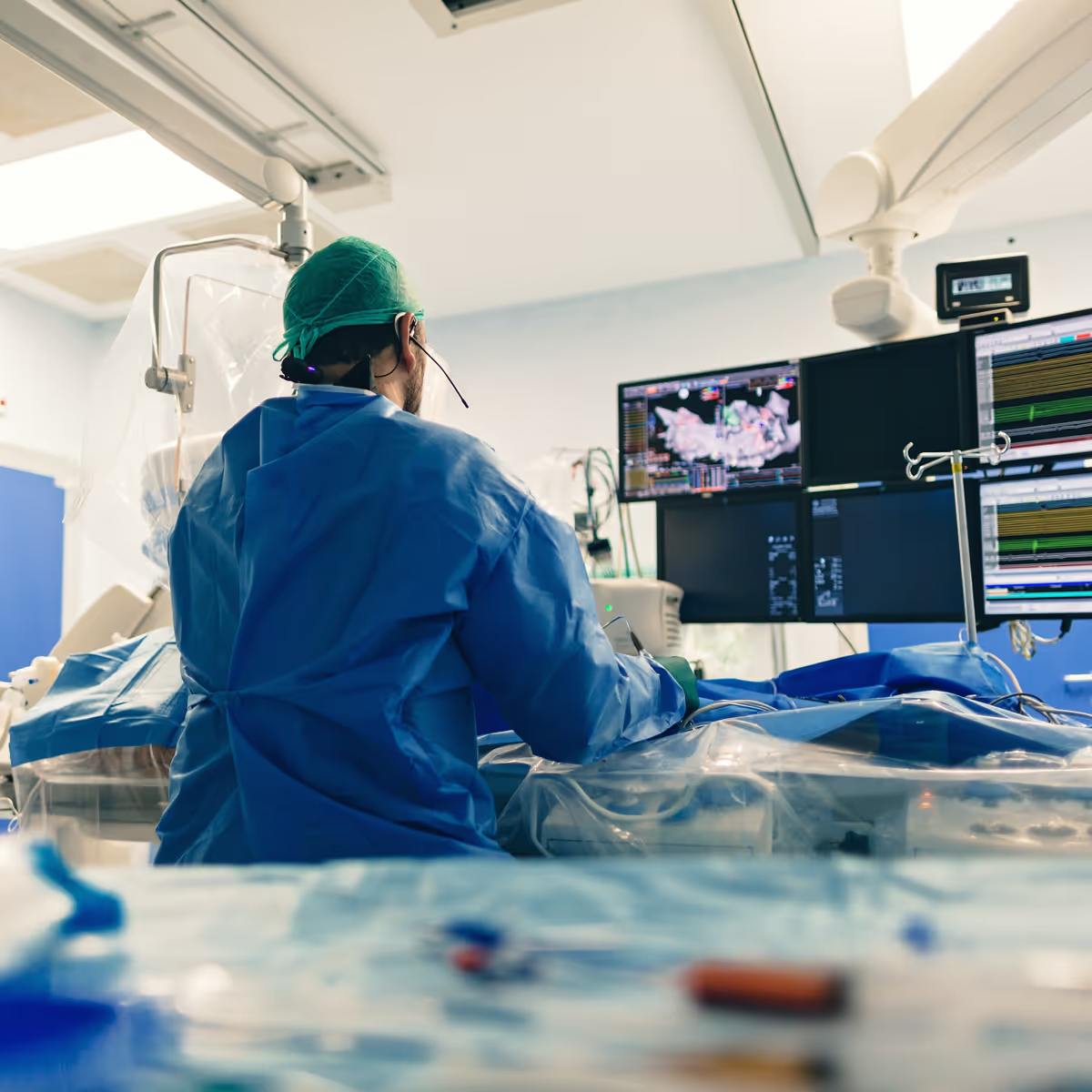

Ablation and energy-based therapies
Energy-based therapies, from RF and ultrasound to cryo and pulse field ablation, are rapidly advancing as safe and effective treatments for arrhythmias.
TTP brings deep expertise in energy delivery, thermal and electrical modelling, and catheter design.
Our teams are driving innovation in non-implant based energy therapies, supported by fusion imaging, robotic navigation, and real-time sensing to guide precise ablation.
By combining these with remote intervention and teleproctoring capabilities, we help create systems that deliver safer, more effective therapies and expand access to care worldwide.


Collaboration that drives results
Partner with us to advance cardiovascular care. At TTP, we combine scientific insight, engineering rigour, and commercial awareness to support every stage of device development - from initial feasibility through to clinical batch manufacturing in line with ISO 13485 and transfer to high-volume manufacture - helping partners create devices that are robust, scalable, and ready for the future of cardiovascular care. Speak to a cardiovascular device development expert today.
How we can help
Enhancing cardiovascular innovation and catheter tracking
TTP develops advanced navigation and tracking technologies that support precise catheter guidance during complex cardiovascular interventions. By combining sensing, modelling and embedded systems, we enable real-time insight into device position and trajectory, helping clinicians navigate challenging vascular pathways with confidence.

Critical structure avoidance
Navigating delicate cardiovascular anatomy requires technologies that help clinicians avoid damaging critical structures such as vessel walls, valves and conduction pathways. TTP develops advanced sensing solutions that provide real-time feedback, supporting safer navigation and more controlled interventions in demanding intravascular environments.

Wireless power and communications for implantable cardiovascular devices
Implantable cardiovascular devices require reliable wireless communication to configure therapies, collect patient data and support device updates. TTP designs wireless power and communication architectures that deliver efficient energy transfer, dependable telemetry and compliance with stringent regulatory requirements.

Catheter and delivery system engineering
Many cardiovascular therapies rely on sophisticated catheter and delivery systems to navigate complex vasculature and accurately deliver treatment. TTP develops advanced catheter technologies, from steerable mechanisms to implant delivery platforms, combining mechanical design and materials expertise to create reliable devices for the cath lab.

Human-centred design and human factors engineering
TTP integrates behavioural science, human factors engineering and multidisciplinary product development to design drug delivery devices that are safe, intuitive and effective in real-world use. From early user research and concept development through formative and summative usability studies, we ensure solutions work across diverse patient populations and care environments, reducing use-related risks while supporting regulatory approval and successful adoption.

Regulatory expertise in medical device development
We have helped hundreds of clients prepare design history files for regulatory submission. Our ISO13485:2015 quality system is audited by BSI every year and several times again by quality auditors from multinational pharma and medical device companies. We speak the language of regulatory and quality teams alongside our deep technical expertise and can help clients bridge the gap between engineering and submission.

Enhancing cardiovascular innovation and catheter tracking
TTP develops advanced navigation and tracking technologies that support precise catheter guidance during complex cardiovascular interventions. By combining sensing, modelling and embedded systems, we enable real-time insight into device position and trajectory, helping clinicians navigate challenging vascular pathways with confidence.

Critical structure avoidance
Navigating delicate cardiovascular anatomy requires technologies that help clinicians avoid damaging critical structures such as vessel walls, valves and conduction pathways. TTP develops advanced sensing solutions that provide real-time feedback, supporting safer navigation and more controlled interventions in demanding intravascular environments.

Wireless power and communications for implantable cardiovascular devices
Implantable cardiovascular devices require reliable wireless communication to configure therapies, collect patient data and support device updates. TTP designs wireless power and communication architectures that deliver efficient energy transfer, dependable telemetry and compliance with stringent regulatory requirements.

Catheter and delivery system engineering
Many cardiovascular therapies rely on sophisticated catheter and delivery systems to navigate complex vasculature and accurately deliver treatment. TTP develops advanced catheter technologies, from steerable mechanisms to implant delivery platforms, combining mechanical design and materials expertise to create reliable devices for the cath lab.

Human-centred design and human factors engineering
TTP integrates behavioural science, human factors engineering and multidisciplinary product development to design drug delivery devices that are safe, intuitive and effective in real-world use. From early user research and concept development through formative and summative usability studies, we ensure solutions work across diverse patient populations and care environments, reducing use-related risks while supporting regulatory approval and successful adoption.

Regulatory expertise in medical device development
We have helped hundreds of clients prepare design history files for regulatory submission. Our ISO13485:2015 quality system is audited by BSI every year and several times again by quality auditors from multinational pharma and medical device companies. We speak the language of regulatory and quality teams alongside our deep technical expertise and can help clients bridge the gap between engineering and submission.

Meet some of the team

Paul Galluzzo

Marco Grasso

Farah Alimagham
Software capability at TTP
Engages in all stages of software and product development, our software capability at TTP covers the full spectrum–from in-depth analysis and system architecture, to prototype design, implementation and development.

Manufacturing capability at TTP
Working seamlessly with our development teams, we take clients’ products through prototype builds, clinical trials, pilot manufacturing and more. Using TTP Manufacturing reduces uncertainty, risk and time to market for our clients.











.avif)