Only a ‘click’ away: click chemistry transforms life science applications
By Wenshu Xu

Click chemistry has come into its own in a wide range of life science applications and opens up opportunities that lie between chemistry, biology and material science, says Wenshu Xu.
Many chemical reactions have traditionally required much control and persuasion – in the form of a wide range of often extreme temperatures, oxygen-free environment, pure starting materials, and downstream purification – and were therefore ill suited to applications in the realm of biology.
Then, twenty years ago, soon-to-be Nobel laureate Barry Sharpless and co-workers began to promote the idea of designing reactions that would simply ‘click’, that is, proceed efficiently and selectively under mild reaction conditions [1].
Initially, these chemists were mainly thinking of clicking together existing organic molecules to accelerate the discovery of new compounds with useful properties. But since then, our ability to customise and manufacture a wide range of biological entities, from antibodies to cells, has grown immeasurably, and click chemistry has also come into its own in a wide range of life science applications.
In this blog, we discuss:
- What is click chemistry?
- Only a ‘click’ away – how to introduce click components
- Click chemistry in life science applications
- Outlook
What is click chemistry?
Click chemistry is modular, wide in scope, gives very high reaction yields and generates only harmless by-products. Ideally insensitive to water and oxygen, the reaction conditions are benign, and the product requires little downstream purification.
This would have excluded classic reactions like the Huisgen cycloaddition of an alkyne and an azide, which is useful for linking molecules, as traditionally this reaction requires refluxing in an organic solvent at 100˚C and still produces a mixture of isomer products.
However, the addition of copper as a catalyst transforms the reaction into a prime example of click chemistry, by speeding up the reaction at lower temperature and favouring the formation of only one product.
Yet, even trace amounts of copper can be difficult to remove and harmful to cells. A copper-free click reaction was created by adding ring strains – akin to ‘loaded springs’ – to the click reactants. Releasing such strain becomes the driving force for the reaction, which readily proceeds at room temperature and without the metal catalyst.
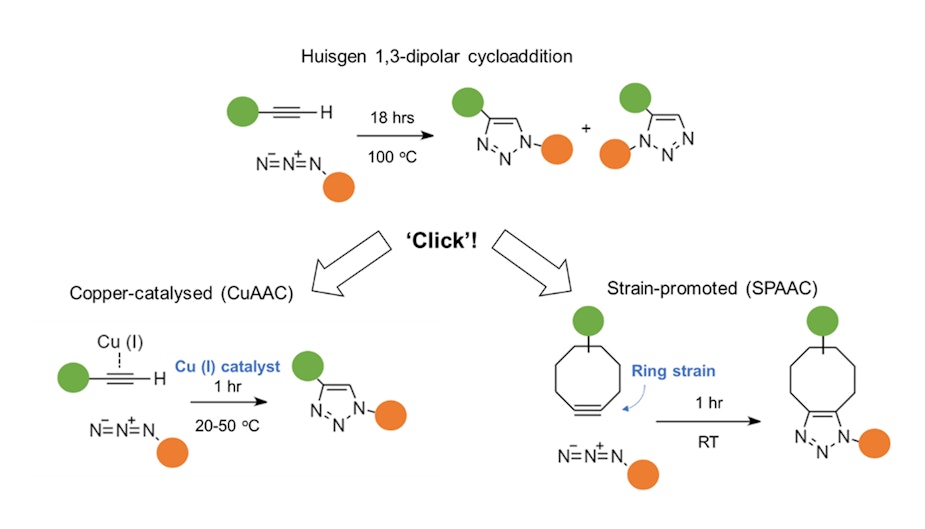
With progress in catalysts and the utilisation of ring strains, many classic chemical reactions have been reinvented as click chemistry and can now be carried out at ambient temperatures and under conditions that are harmless to living organisms and biological molecules.
These attributes make click chemistry particularly appealing for bioconjugation, where biocompatible, highly specific and non-toxic chemical strategies are required to join together two or more molecules, one of which is a biomolecule.
Only a ‘click’ away
Similar to a lock and key system, click moieties are designed to react only with their click counterparts without cross-reaction with other functional groups.
As they are small as well as water soluble, clickable reagents are also easily taken up by cells and can even cross the blood-brain barrier. Clickable components can be incorporated into the building blocks of cells, proteins and nucleic acids via biosynthesis or chemical modifications.
For example, during protein biosynthesis, cells may be tricked into incorporating an unnatural amino acid containing a click-ready group into the protein sequence, by a combination of specially engineered tRNA, ribosome and parallel genetic codes [2].
Alternatively, one can directly modify features of biomolecules, such as the glycan chains on antibodies and on the cell surface, to functionalise them with click-ready groups.
Thereafter, these click moiety-containing biologics can be coupled to various other probes such as fluorescent labels, small-molecule drugs, carbohydrates, DNAs or RNAs, and proteins.
The good news for non-chemists is that there are already a number of reagent companies that sell ready-made click building blocks with easy-to-follow recipes, so they can be used in the desired applications straightaway. This provides a solution for carrying out simple chemistry on bench, without having to access Schlenk line, fume hood, and chromatography in a chemical laboratory [3, 4, 5].
Click chemistry in life science applications
Click chemistry is already widely used in pharmaceutical and life science applications and increasingly prompting large commercial deals.
Drug discovery has been boosted by click chemistry, as not only the resulting chemical linkages (such as the triazole ring) are highly useful in medicinal chemistry, but also new and potentially more active compounds can be formed by clicking existing compounds together [6]. This is seen in fragment-based drug screening and PROTAC, a targeted protein degradation drug platform, which is considered as the next hot drug development area [7].
In the nucleic acid sequencing arena, ClickSeq, a click chemistry-based method for RNA sequencing has proven to be more robust and less error-prone than the enzymatic ligation commonly used to link sequencing adaptors during the creation of libraries for next-generation Illumina sequencing [8].
Click chemistry also enables the coupling of antibodies and cytotoxic drugs to produce site-specific and stable antibody-drug conjugates (ADCs) [9]. In 2019, AstraZeneca signed a $6.9 billion deal with Daiichi Sankyo to develop ADCs to replace Herceptin for the treatment of HER2 positive breast cancer [10].
In a previous blog, we have discussed how we used click chemistry to fabricate stable nanoparticle conjugates for detecting biomarkers on cells.
Outlook
Many other applications of click chemistry are in the pipeline of pre-commercial research.
In life science research, click chemistry is used for in vivo fluorescent labelling of proliferating cells in live experimental organisms: injected clickable nucleosides are incorporated into the replicating DNA and a click counterpart with fluorescent label is used to visualise cell proliferation.
For example, zebrafish is often used as a model to study seizures and to understand how brain tissue responds to recurrent seizures and find new treatment paradigms in humans. In vivo labelling with the help of click chemistry has been used to investigate the relationship between recurrent seizures and cellular response [11].
Click chemistry can also be used to create new biomaterials by conjugating whole living cells to hydrogels. Wound healing and tissue engineering benefit from live functional hydrogels made by crosslinking clickable cells with polymers. The hydrogel can be formed in situ once mixed with these cells, providing a scaffold for them to grow, and, in turn, the cells promote better adhesion of hydrogel to the surface at the site of interest. One study has shown that this approach helps to restore muscle strength in genetically defective mice [12].
Click chemistry provides us with a simple yet powerful toolbox for innumerable biological applications. Its utility is expanding rapidly in areas such as drug development, next-generation sequencing, dynamic in vivo imaging and biomaterial preparation for regenerative medicines. Click chemistry opens up many further opportunities that lie between chemistry, biology and material science.
Diagnostics | Life Science | Medical Imaging | Digital Health | Biosensors | Healthcare Blogs | Healthcare Case Studies